

| So light is a
stream of photons! But why then does it act like a wave? Why do we observe diffraction and interference? |
|
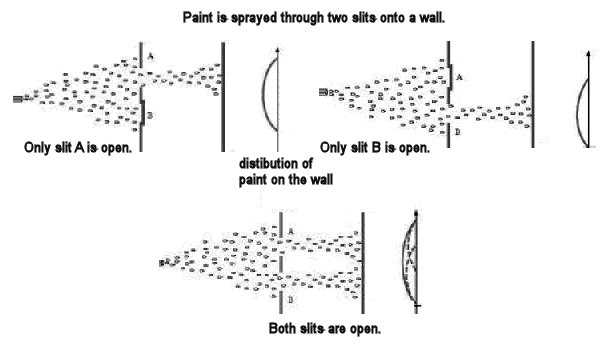 |
The photons definitely do not behave like macroscopic particles, even very small macroscopic particles. If we positioned a mask with two slits in front of a wall and used a paint gun to spray paint through the slits onto the wall, we would not expect the paint to produce an interference pattern on the wall. We would expect to observe two sharp lines of paint on the wall, images of the two slits through which the paint was sprayed. Even if we made the slits as narrow as possible and put them as close together as possible for the paint to still pass through them without clogging them up, we would not expect to observe an interference pattern. The figure below shows the pattern we expect when paint is sprayed through two closely spaced slits. |
 |
If we shine light onto a screen through a mask with two large holes, whose dimensions and separation are much bigger than the wavelength of light, we expect to observe sharp shadows of the edges of the holes on the screen. But as we decease the size and separation of the holes, the shadows begin to wash out and an interference pattern appears. That pattern is a characteristic signature of a wave and is shown in the figure below. |
So what is light, an electromagnetic wave or a stream of photons? What is our current understanding of the nature of light?
Quantum mechanics views photons as quanta or packets of energy. But these quanta behave nothing like macroscopic particles.
In a single or double slit experiment, we can only predict the probability that the photon will strike the screen in a given spot.
That probability can be calculated using the wave equation for electromagnetic waves.
Where that equation predicts a high light intensity, the probability is large, and where it predicts a low light intensity, the probability is small.
In other words, where the wave equation predicts constructive interference, the photon is likely to be detected, and where it predicts destructive interference, the probability of detecting the photon is small or zero
Each photon of visible light has only a small amount of energy, and our eyes are not sensitive enough to detect a single photon. So we only see something, when many photons hit the retina at approximately the same time. We see a continuous distribution of photons, a light pattern.
But there are detectors with enough sensitivity to detect individual photons of visible light. What does such a detector "see" when it replaces the screen in a double-slit experiment with the light intensity turned way down? It "sees" packets of energy arriving at well defined positions. When only a few photons have been counted, the positions seem to be randomly distributed. But if we keep a record of the arrival positions, or if many photons arrive in a small time interval, the double slit interference pattern emerges.
Demonstration:
 |
We will observe two-slit interference, one photon at a time using an apparatus made by TeachSpin. |
The linked spreadsheet lets you simulate a double slit experiments using a detector that can detect individual photons. The two slits are 50 micrometer wide and 150 micrometer apart. The distance from the slits to the screen is 1 m. The pattern is a product of a single slit and a double slit diffraction pattern. Individual photons arrive at the screen. After many photons have arrived, the interference pattern emerges. You can vary the wavelength and intensity of the incident light.
How is it possible for light to propagate as if it were a wave and yet to be detected as if it were a particle? How can a single particle interfere with itself? This paradox is the central theme in Richard Feynman's introduction to the fundamentals of quantum mechanics:
"We choose to examine a phenomenon which is impossible, absolutely impossible, to explain in any classical way, and which has in it the heart of quantum mechanics. In reality, it contains the only mystery. We cannot make the mystery go away by explaining how it works . . . In telling you how it works we will have told you about the basic peculiarities of all quantum mechanics."
Photons are particles of light. But they do not behave like macroscopic particles. We cannot use the laws of classical physics to predict the behavior of individual photons. We have to use the rules of quantum mechanics. Quantum mechanics often makes probabilistic predictions. The probabilities can be calculated using a wave equation.
The photons of an electromagnetic monochromatic (monochromatic = single color or wavelength) EM wave,
E(x,t) = Emaxcos(kx - ωt + φ) = Emaxcos((2π/λ)x - (2πf)t + φ),
all have energy E = hf = hc/λ and momentum p = hf/c = h/λ.
For such an EM wave the energy and momentum of each individual photon is predicted exactly. But the wave described by this equation has infinite extend, and all photons of this wave are exactly alike, they are indistinguishable. We cannot predict where we will find any particular photon at any specific time.
To track individual photons or groups of photons, the EM wave we use predicting their behavior must be a wave pulse or wave packet. A wave packet has finite extend, and the photons will be found in regions where the wave packet does not have zero amplitude. But a wave packet must be build from harmonic waves with many different frequencies or wavelength. We therefore do not exactly know the energy E = hf and momentum p = h/λ of the individual photons in the wave packet. Only waves that extend forever in space and time have a precisely defined wavelength and frequency.
We write Δx Δk ~ 1.
We write Δt Δω ~ 1.
Since for photons the energy is E = hf and momentum is p = h/λ, we cannot precisely know the energy and momentum of the individual photons in the wave packet. Rewriting k = 2π/λ = (2π/h)p, we find
Δx Δp ~ h/2π.
This is the famous Heisenberg uncertainty principle. Here we are applying it to a photon. For a photon, we cannot predict its position and momentum with absolute certainty. The product of the uncertainties is on the order of h/2π or greater. The quantity h/2π is often denoted by ћ (hbar), ћ = 1.054*10-34 Js.
Rewriting ω = 2πf = (2π/h)E, we find
Δt ΔE ~ h/2π.
This is another form of the uncertainty principle. If there is an uncertainty in the observation time, there will be an uncertainty in the energy.